The cosmetic registration process needs strategic planning and a review of the ingredients in the cosmetic products and labeling for registration in India. The cosmetic companies intending to import and market cosmetic products in India have to go through this tedious process. The cosmetic application should adequately define the products in the respective categories.
In this article, let’s take a look at steps of cosmetic registration process in India.
STEP 1: CREATION OF SUGAM LOGIN CREDENTIALS
Various documents uploaded on SUGAM online portal of CDSCO for Importers’ registration
- Affidavit / Undertaking signed & stamped by the authorized signatory and send us color scanned copy of the same
- Cover Letter signed & stamped by the authorized signatory
- Aadhar Card Copy/Passport copy of importer.
- IEC Certificate
- Additional Information Sheet: Information required to fill Sugam details
After uploading, hard copy of these documents is sent to CDSCO for verification.
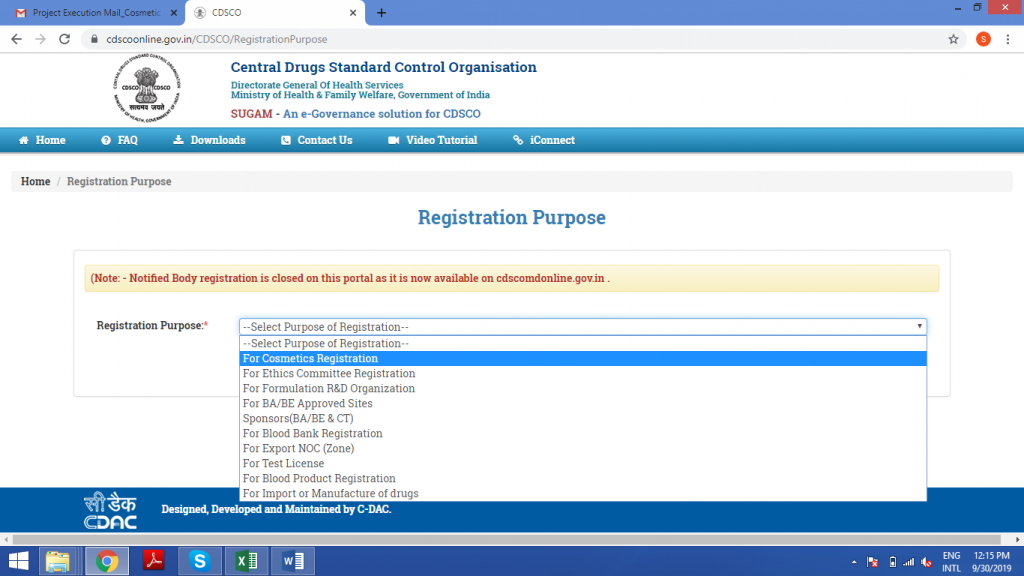
Select the purpose of registration as ‘For Cosmetic Registration’.
STEP 2: DRAFTING THE FINAL DOCUMENTS FOR COSMETIC REGISTRATION
- List of Ingredients for each product, Labels (artwork), certificate of analysis and complete product list
- Additional information
- Name and Complete address of Manufacturer with Telephone number, Fax and Email ID
- Net Content (Pack size of products)
- Copy of Free Sale Certificate: It must include the products name, with the name of the brand preceding the product’s name, along with names of variants.
- Valid copy of manufacturing license
- Documents from Manufacturer
- Power of Attorney
- Testing Protocol
- Undertaking for microbial tests
- Undertaking for Fragrance
- Undertaking for Animal Testing
- Undertaking for Heavy Metals and Hexachlorophene
STEP 3: UPLOAD THE FINAL DOCUMENTS
The sugam portal will be activated now. Log in to the account using the User ID and Password.
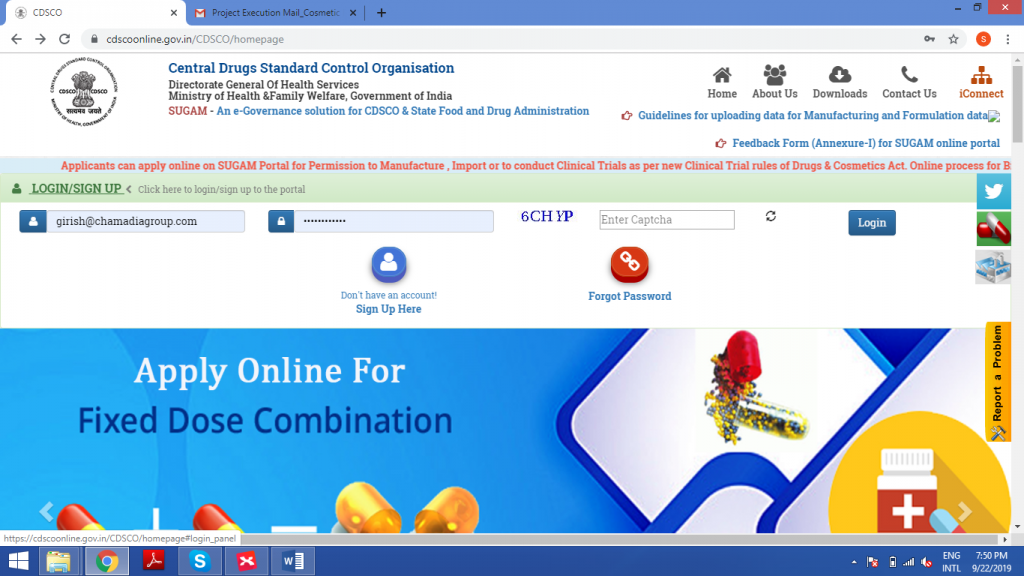
STEP 4: FORM SUBMISSION
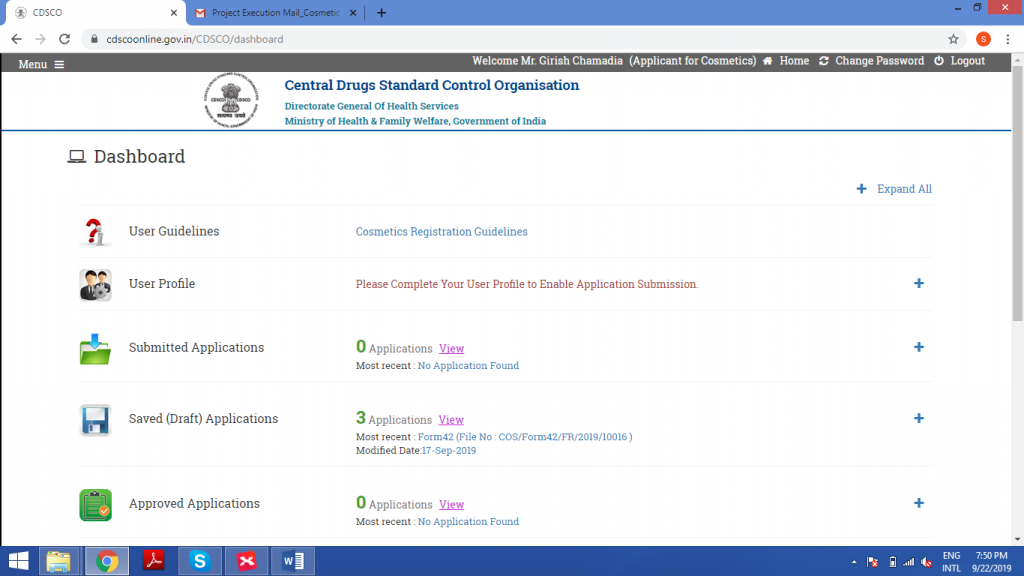
This is what the dashboard will look like once the Log-in is successful. Click on Menu.
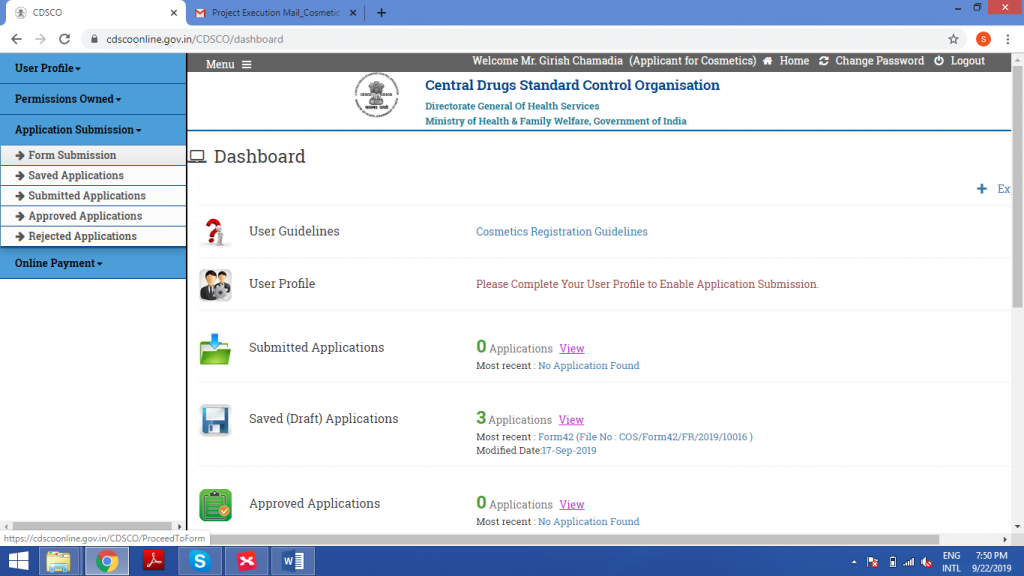
Under Application Submission, click on Form Submission.
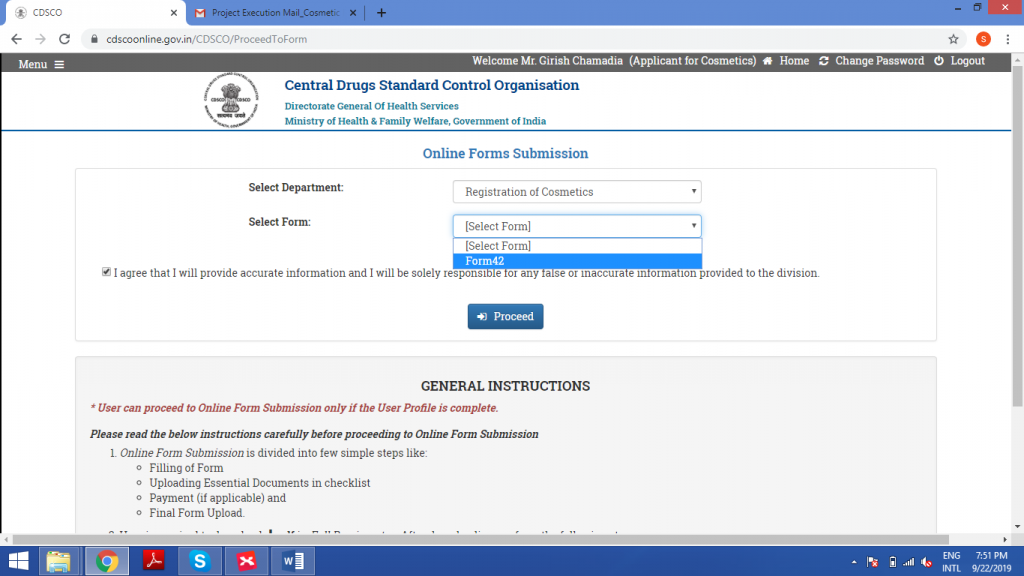
Select ‘Registration of Cosmetics’ under Department, then select Form42.
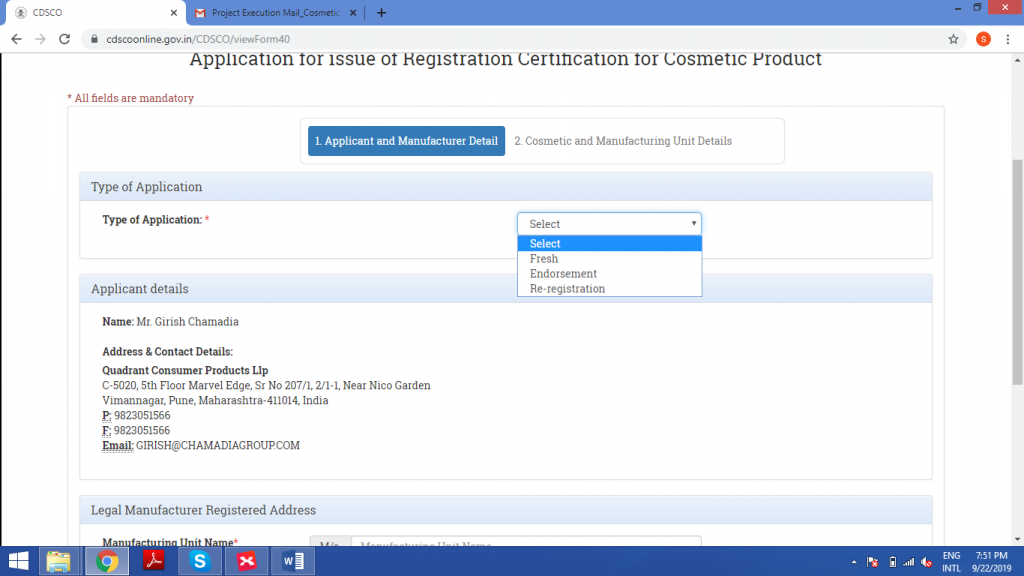
Select the type of application, according to the specifications of the project.
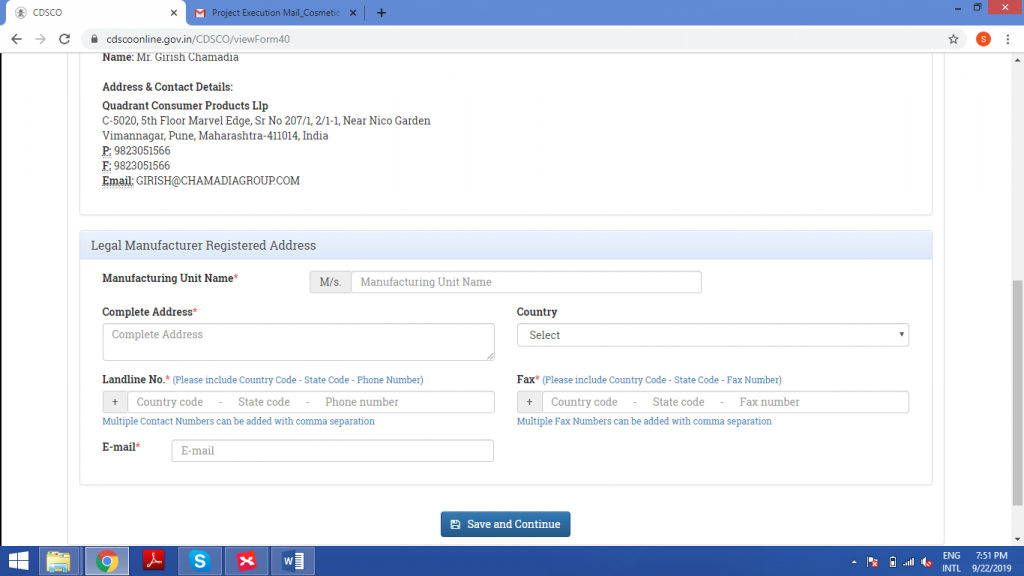
Fill out the details of the manufacturer, precisely. After filling these details, cross-check them with the documents once, to make sure that no error has been made.
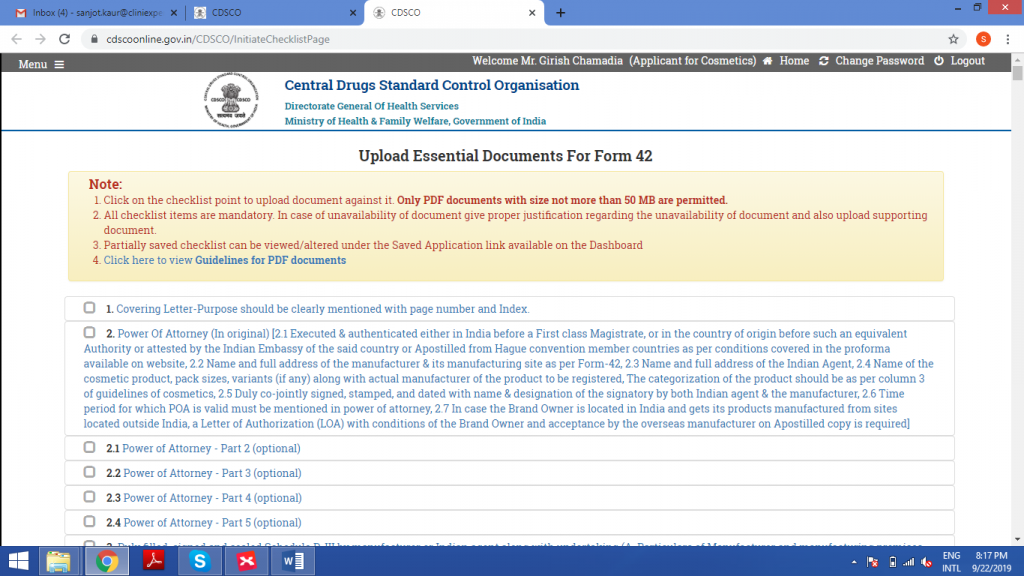
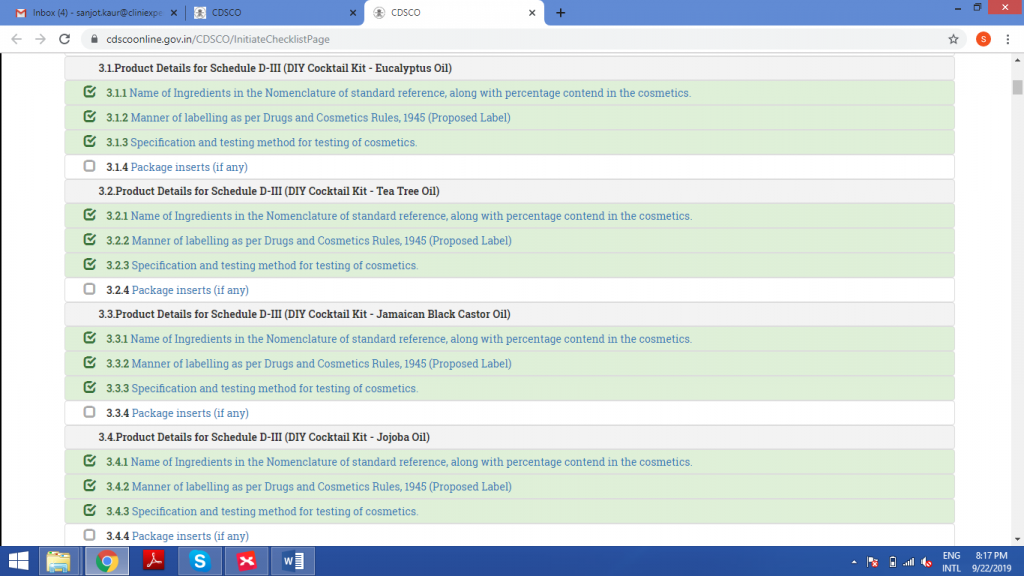
STEP 5: GENERATION OF CHECKLIST
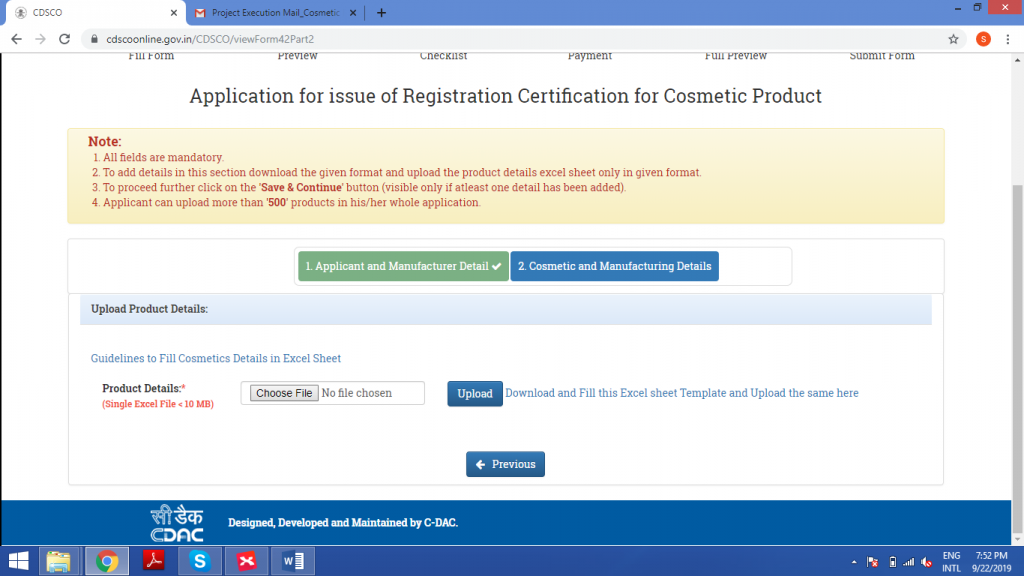
Upload the excel sheet in this field.
Check all the details in the next step.
Once the application is submitted, it cannot be edited further.
Preview of Form42 is generated.
STEP 6: PAYMENT OF GOVERNMENT FEE THROUGH BHARATKOSH FOR COSMETIC PRODUCTS
The process of payment is:
- Enter login details on SUGAM portal
- The screen will now display the dashboard. Click on Switch role. Go to Indian Agent. Click on Menu, then click on Online Payment. Next, click on make payment.
- Fill all the fields (e.g. Division name, Purpose, amount etc.) on the page titled ‘Online Challan Payment Details. Click on submit. Then click on the link.
- Check all the details and after confirming them, click on ‘Confirm’ and the payment gateway will open. After making the payment, click on “View Payment Status”
- Click on “Download Challan from BharatKosh” and fill all the details (OTP will be sent to mobile number and E-Mail mentioned on SUGAM profile). Download challan. Challan will be generated along with the receipt which must be saved.
STEP 7: GENERATION OF FORM42
- Once the documents are submitted, the challan must be uploaded.
- Form42 will be generated.
- Download the PDF file and send it across to the applicant (Indian Agent) to get it signed and stamped.
STEP 8: SUBMISSION OF APPLICATION
- Upload signed and stamped copy of Form42.
- Note application/file number for reference.
References:
- pdf. Available at: https://cdsco.gov.in/opencms/export/sites/CDSCO_WEB/Pdf-documents/cosmetics/Guidancedoccos.pdf .Accessed on 21st October, 2020.
- pdf. Available at: https://cdsco.gov.in/opencms/export/sites/CDSCO_WEB/Pdf-documents/SUGAM_user_manual.pdf . Accessed on 21st October, 2020.


